The filling of the 2p orbital places eight electrons in the second shell, a "stable octet". The order of filling will place one electron in each before placing two in any one orbital since the mutually repulsive electrons prefer to be further apart. The three possibilities for 2p orbitals can be associated with spatial directions, say x,y and z. The elements from boron to neon in the periodic table are filling the 2p orbital. It tends to lose that electron to revert to the stable helium electron structure. Lithium is a very active alkali metal, having just one electron in the second shell. With two electrons in the 1s state, it is filled and stable, forming the noble gas helium. 
The 1s spatial state can have two states of electron spin, which we usually just refer to as "spin up" and "spin down". The buildup process for the first 12 elements of the periodic table gives some hints about the origin of chemical reactivity. Since all of nature tends to the lowest energy state, the individual states will fill in the order of ascending energy. The Pauli exclusion principle tightly constrains the entire buildup process, allowing only one electron to occupy each available state. Perhaps a better way to keep track of the buildup process is to just track the filling of the states, emphasizing the order of buildup and the fact that the states are definite, quantized energy states. The division into main shells encourages a kind of "planetary model" for the electrons, and while this is not at all accurate as a description of the electrons, it has a certain mnemonic value for keeping track of the buildup of heavier elements. This spreading eventually leads to overlap, with the 4s sub-level being lower in energy than the 3d sub-level. But in larger atoms, the energy depends also upon the orbital quantum number so the sub-levels are filled in the order s, p, d, f, etc. This is strictly true for the hydrogen atom where the energy levels depend only upon the principal quantum number (fine structure neglected). In general terms, the higher shells have higher energy (less tightly bound) and are on the average further out from the nucleus. The letters designate the sub-shells and follow the historical spectroscopic notation. The numbers of the main shells, like 1s, arise from the principal quantum number n in the quantum mechanical description of the electrons. 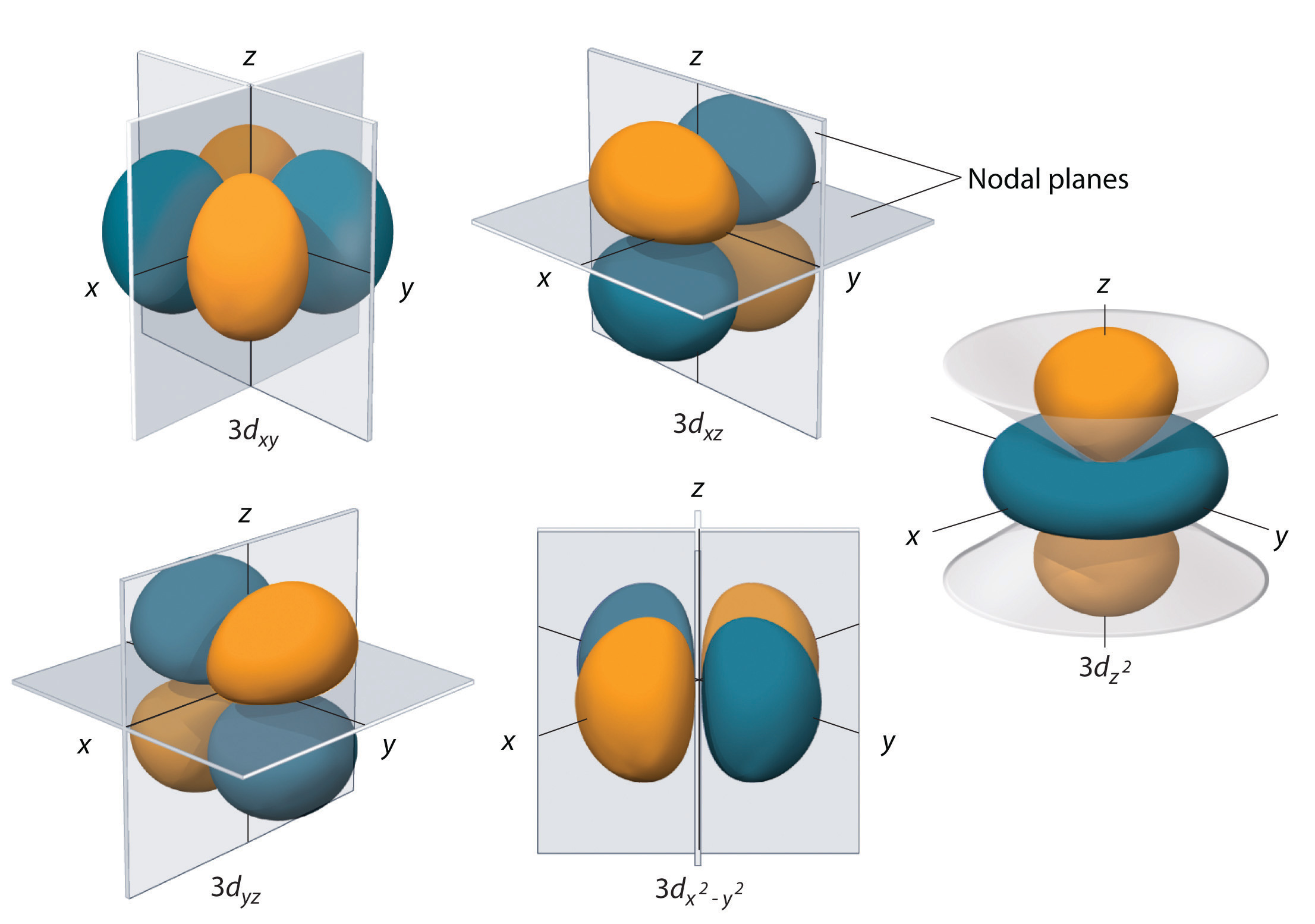
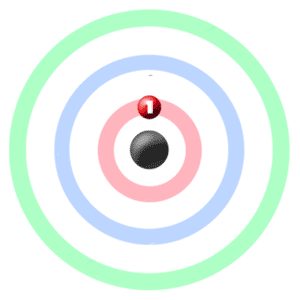
These sketches arise from the hydrogen wavefunctions which map the electron density. The limits on the occupation of the subshells arise from the quantum numbers for the atomic electrons and their relationship to each other. Rough sketches of the electron density for the first three shells of the hydrogen atom can give an impression of the constraints that govern the buildup of the periodic table. Similarly, a negative Δ E means that the atom has lost energy through emission of a photon.Ĭonservation of energy requires that the energy of the photon, E photon = hc/λ, equals the absolute value of the energy difference, |Δ E|, for emission or absorption.Visualizing Electron Orbitals Visualizing Electron Orbitals The energy of a photon, E photon, can be determined from either its frequency or its wavelength:Į_ \right)Ī positive Δ E means that the atom’s energy increased, corresponding to absorption of a photon: the photon’s energy has been added to the atom’s initial energy. In a vacuum, electromagnetic radiation travels at the speed of light ( c): λ ν = c = 2.998 × 10 8 m sĮlectromagnetic radiation occurs in small, indivisible quantities of energy called photons. The oscillating fields (waves) are characterized by wavelength (λ, measured in meters, m) and frequency (\nu, measured in hertz, Hz or s −1). We will ask you to refer back to what you have written when you complete this section.Įlectromagnetic radiation consists of oscillating, perpendicular electric and magnetic fields that travel through space and can transfer energy. If there is anything you remember being puzzled about, write that down as well. Also write what you recall about the relation of spectra to atomic structure-how electrons are arranged in atoms. After the heading write down what you remember about atomic spectra from courses you have already taken. In your course notebook, make a heading for Atomic Spectra and Atomic Structure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
